State-of-the art pMHC stability assessment
Screen peptides or peptide libraries for optimal MHC binders. Our stability assay measures the dissociation of pMHC complexes in real-time at 37°C allowing you to discriminate between peptides that support the formation of stable (more likely to be immunogenic) or unstable (less likely to be immunogenic) peptide-MHC complexes.
The stability assay is ideally suited to further stratify peptide targets predicted to bind to MHC, whether the goal is to identify new vaccine targets or to screen protein drug candidates for harboring potential immunogenic fragments. More than 90 HLA allotypes can be assayed for pMHC stability.
Assessment of peptide-MHC complex stability is an important parameter when screening for immunogenic peptides. The stability of any given peptide-MHC complex can be determined by monitoring the dissociation of immune complexes at 37°C and calculate the half-life. The half-life of a peptide-MHC complex have been demonstrated to be a better correlate of immunogenicity than the corresponding peptide affinity for the MHC (Harndahl et.al1).
We have developed a high-throughput assay to monitor the dissociation of peptide MHC class I complexes (figure below) and determine the half-life of any peptide. Our stability measurement service applies to all MHC class I molecules available and can assist your epitope / neo-epitop screening.
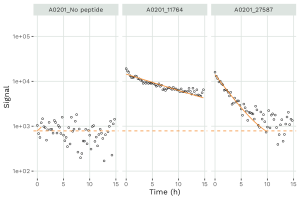
The determination of the half-life of a set of peptides (pairwise matched for HLA-A*02:01 affinity) previously described as being either immunogenic or non-immunogenic (Assarsson E, et. al2) revealed that verified T cell epitopes produced significantly longer half-lives compared to verified non-immunogenic peptides. A ROC analysis of the half-lives in the two groups produced an AUC of 0.97 suggesting that stability is a key parameter for immunogenicity assessments.
Bibliography
- Harndahl, M., M. Rasmussen, G. Roder, I. Dalgaard Pedersen, M. Sorensen, M. Nielsen and S. Buus (2012). “Peptide-MHC class I stability is a better predictor than peptide affinity of CTL immunogenicity.” Eur J Immunol 42(6): 1405-1416.
- Assarsson E1, Sidney J, Oseroff C, Pasquetto V, Bui HH, Frahm N, Brander C, Peters B, Grey H, Sette A. “A quantitative analysis of the variables affecting the repertoire of T cell specificities recognized after vaccinia virus infection.” J Immunol. 2007 Jun 15;178(12):7890-901.